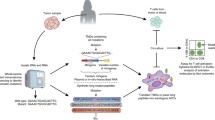
Abstract
Although cancer immunotherapy shows efficacy with adoptive T cell therapy (ACT) and antibody-based immune checkpoint blockade, efficacious therapeutic vaccination of cancer patients with tumor-associated antigens (TAAs) remains largely unmet. Current cancer vaccines utilize nonmutated shared TAAs that may have suboptimal immunogenicity. Experimental evidence underscores the strong immunogenicity of unique TAAs derived from somatically mutated cancer proteins, whose massive characterization has been precluded until recently by technical limitations. The development of cost-effective, high-throughput DNA sequencing approaches makes now possible the rapid identification of all the somatic mutations contained in a cancer cell genome. This method, combined with robust bioinformatics platforms for T cell epitope prediction and established reverse immunology approaches, provides us with an integrated strategy to identify patient-specific unique TAAs in a relatively short time, compatible with their potential use in the clinic. Hence, it is now for the first time possible to quantitatively define the patient’s unique tumor antigenome and exploit it for vaccination, possibly in combination with ACT and/or immune checkpoint blockade to further increase immunotherapy efficacy.
Similar content being viewed by others

Abbreviations
- ACT:
-
Adoptive T cell therapy
- Ags:
-
Antigens
- CAN-genes:
-
Candidate cancer genes
- exome-Seq:
-
Exome sequencing
- mAb:
-
Monoclonal antibody
- MSI:
-
Microsatellite instable
- MSS:
-
Microsatellite stable
- RNA-Seq:
-
RNA sequencing
- SNP-array:
-
Single nucleotide polymorphism
- TAAs:
-
Tumor-associated antigens
- TCR:
-
Tell receptor
- TCGA:
-
The Cancer Genome Atlas
- Tregs:
-
CD4+CD25+T regulatory cells
- UV:
-
Ultraviolet
References
Coulie PG, Van den Eynde BJ, van der Bruggen P, Boon T (2014) Tumour antigens recognized by T lymphocytes: at the core of cancer immunotherapy. Nature Rev Cancer 14:135–146
Gilboa E (1999) The makings of a tumor rejection antigen. Immunity 11:263–270
Novellino L, Castelli C, Parmiani G (2005) A listing of human tumor antigens recognized by T cells: March 2004 update. Cancer Immunol Immunother 54:187–207
Parmiani G, Castelli C, Dalerba P, Mortarini R, Rivoltini L, Marincola FM, Anichini A (2002) Cancer immunotherapy with peptide-based vaccines: what have we achieved? Where are we going? J Natl Cancer Inst 94:805–818
Rosenberg SA, Yang JC, Restifo NP (2004) Cancer immunotherapy: moving beyond current vaccines. Nat Med 10:909–915
Speiser DE, Lienard D, Rufer N, Rubio-Godoy V, Rimoldi D, Lejeune F, Krieg AM, Cerottini JC, Romero P (2005) Rapid and strong human CD8+ T cell responses to vaccination with peptide, IFA, and CpG oligodeoxynucleotide 7909. J Clin Invest. 115:739–746
Kantoff PW, Higano CS, Shore ND et al (2010) Sipuleucel-T immunotherapy for castration-resistant prostate cancer. N Engl J Med 363:411–422
Rivoltini L, Canese P, Huber V et al (2005) Escape strategies and reasons for failure in the interaction between tumour cells and the immune system: how can we tilt the balance towards immune-mediated cancer control? Expert Opin Biol Ther. 5:463–476
Savage PA, Leventhal DS, Malchow S (2014) Shaping the repertoire of tumor-infiltrating effector and regulatory T cells. Immunol Rev 259:245–258
Mortarini R, Piris A, Maurichi A et al (2003) Lack of terminally differentiated tumor-specific CD8+ T cells at tumor site in spite of antitumor immunity to self-antigens in human metastatic melanoma. Cancer Res 63:2535–2545
Hailemichael Y, Dai Z, Jaffarzad N et al (2013) Persistent antigen at vaccination sites induces tumor-specific CD8(+) T cell sequestration, dysfunction and deletion. Nat Med 19:465–472
Prehn RT, Main JM (1957) Immunity to methylcholanthrene-induced sarcomas. J Natl Cancer Inst 18:769–778
Klein G, Sjogren HO, Klein E, Hellstrom KE (1960) Demonstration of resistance against methylcholanthrene-induced sarcomas in the primary autochthonous host. Cancer Res 20:1561–1572
Mumberg D, Wick M, Schreiber H (1996) Unique tumor antigens redefined as mutant tumor-specific antigens. Semin Immunol 8:289–293
Lurquin C, Van Pel A, Mariame B, De Plaen E, Szikora JP, Janssens C, Reddehase MJ, Lejeune J, Boon T (1989) Structure of the gene of tum- transplantation antigen P91A: the mutated exon encodes a peptide recognized with Ld by cytolytic T cells. Cell 58:293–303
Foley EJ (1953) Antigenic properties of methylcholanthrene-induced tumors in mice of the strain of origin. Cancer Res 13:835–837
Srivastava P (2002) Interaction of heat shock proteins with peptides and antigen presenting cells: chaperoning of the innate and adaptive immune responses. Annu Rev Immunol 20:395–425
Srivastava PK, Duan F (2013) Harnessing the antigenic fingerprint of each individual cancer for immunotherapy of human cancer: genomics shows a new way and its challenges. Cancer Immunol Immunother 62:967–974
Castelli C, Ciupitu AM, Rini F, Rivoltini L, Mazzocchi A, Kiessling R, Parmiani G (2001) Human heat shock protein 70 peptide complexes specifically activate antimelanoma T cells. Cancer Res 61:222–227
Rivoltini L, Castelli C, Carrabba M et al (2003) Human tumor-derived heat shock protein 96 mediates in vitro activation and in vivo expansion of melanoma- and colon carcinoma-specific T cells. J Immunol. 171:3467–3474
Mazzaferro V, Coppa J, Carrabba MG et al (2003) Vaccination with autologous tumor-derived heat-shock protein gp96 after liver resection for metastatic colorectal cancer. Clin Cancer Res 9:3235–3245
Parmiani G, Testori A, Maio M et al (2004) Heat shock proteins and their use as anticancer vaccines. Clin Cancer Res 10:8142–8146
Wolfel T, Hauer M, Schneider J et al (1995) A p16INK4a-insensitive CDK4 mutant targeted by cytolytic T lymphocytes in a human melanoma. Science 269:1281–1284
Coulie PG, Lehmann F, Lethe B, Herman J, Lurquin C, Andrawiss M, Boon T (1995) A mutated intron sequence codes for an antigenic peptide recognized by cytolytic T lymphocytes on a human melanoma. Proc Natl Acad Sci USA 92:7976–7980
Stratton MR, Campbell PJ, Futreal PA (2009) The cancer genome. Nature 458:719–724
Lawrence MS, Stojanov P, Polak P et al (2013) Mutational heterogeneity in cancer and the search for new cancer-associated genes. Nature 499:214–218
Lawrence MS, Stojanov P, Mermel CH et al (2014) Discovery and saturation analysis of cancer genes across 21 tumour types. Nature 505:495–501
Vogelstein B, Papadopoulos N, Velculescu VE, Zhou S, Diaz LA Jr, Kinzler KW (2013) Cancer genome landscapes. Science 339:1546–1558
Margulies M, Egholm M, Altman WE et al (2005) Genome sequencing in microfabricated high-density picolitre reactors. Nature 437:376–380
Thomas RK, Baker AC, Debiasi RM et al (2007) High-throughput oncogene mutation profiling in human cancer. Nat Genet 39:347–351
Garraway LA, Lander ES (2013) Lessons from the cancer genome. Cell 153:17–37
Cancer Genome Atlas Network (2012) Comprehensive molecular characterization of human colon and rectal cancer. Nature 487:330–337
Dudley ME, Roopenian DC (1996) Loss of a unique tumor antigen by cytotoxic T lymphocyte immunoselection from a 3-methylcholanthrene-induced mouse sarcoma reveals secondary unique and shared antigens. J Exp Med 184:441–447
Lennerz V, Fatho M, Gentilini C, Frye RA, Lifke A, Ferel D, Wolfel C, Huber C, Wolfel T (2005) The response of autologous T cells to a human melanoma is dominated by mutated neoantigens. Proc Natl Acad Sci USA 102:16013–16018
Anichini A, Mortarini R, Maccalli C, Squarcina P, Fleischhauer K, Mascheroni L, Parmiani G (1996) Cytotoxic T cells directed to tumor antigens not expressed on normal melanocytes dominate HLA-A2.1-restricted immune repertoire to melanoma. J Immunol. 156:208–217
Parmiani G, De Filippo A, Novellino L, Castelli C (2007) Unique human tumor antigens: immunobiology and use in clinical trials. J Immunol. 178:1975–1979
Robbins PF, Lu YC, El-Gamil M et al (2013) Mining exomic sequencing data to identify mutated antigens recognized by adoptively transferred tumor-reactive T cells. Nat Med 19:747–752
van Rooij N, van Buuren MM, Philips D et al (2013) Tumor exome analysis reveals neoantigen-specific T-cell reactivity in an ipilimumab-responsive melanoma. J Clin Oncol 31:439–442
Tran E, Turcotte S, Gros A et al (2014) Cancer immunotherapy based on mutation-specific CD4+ T Cells in a patient with epithelial cancer. Science 344:641–645
Matsushita H, Vesely MD, Koboldt DC et al (2012) Cancer exome analysis reveals a T-cell-dependent mechanism of cancer immunoediting. Nature 482:400–404
Thornton AM, Shevach EM (2000) Suppressor effector function of CD4 + CD25+ immunoregulatory T cells is antigen nonspecific. J Immunol. 164:183–190
von Boehmer H (2005) Mechanisms of suppression by suppressor T cells. Nat Immunol 6:338–344
Bui JD, Uppaluri R, Hsieh CS, Schreiber RD (2006) Comparative analysis of regulatory and effector T cells in progressively growing versus rejecting tumors of similar origins. Cancer Res 66:7301–7309
Pardoll DM (2012) The blockade of immune checkpoints in cancer immunotherapy. Nature Rev Cancer. 12:252–264
Hodi FS, O’Day SJ, McDermott DF et al (2010) Improved survival with ipilimumab in patients with metastatic melanoma. N Engl J Med 363:711–723
Topalian SL, Hodi FS, Brahmer JR et al (2012) Safety, activity, and immune correlates of anti-PD-1 antibody in cancer. N Engl J Med 366:2443–2454
Thomas RK, Nickerson E, Simons JF et al (2006) Sensitive mutation detection in heterogeneous cancer specimens by massively parallel picoliter reactor sequencing. Nat Med 12:852–855
Zhang Q, Wang P, Kim Y et al (2008) Immune epitope database analysis resource (IEDB-AR). Nucleic Acids Res 36:W513–W518
Lundegaard C, Lamberth K, Harndahl M, Buus S, Lund O, Nielsen M (2008) NetMHC-3.0: accurate web accessible predictions of human, mouse and monkey MHC class I affinities for peptides of length 8-11. Nucleic Acids Res 36:W509–W512
Nielsen M, Lundegaard C, Blicher T, Peters B, Sette A, Justesen S, Buus S, Lund O (2008) Quantitative predictions of peptide binding to any HLA-DR molecule of known sequence: NetMHCIIpan. PLoS Comput Biol 4:e1000107
Castle JC, Kreiter S, Diekmann J et al (2012) Exploiting the mutanome for tumor vaccination. Cancer Res 72:1081–1091
Rajasagi M, Shukla SA, Fritsch EF et al (2014) Systematic identification of personal tumor-specific neoantigens in chronic lymphocytic leukemia. Blood 124:453–462
Segal NH, Parsons DW, Peggs KS, Velculescu V, Kinzler KW, Vogelstein B, Allison JP (2008) Epitope landscape in breast and colorectal cancer. Cancer Res 68:889–892
Fritsch EF, Rajasagi M, Ott PA, Brusic V, Hacohen N, Wu CJ (2014) HLA-binding properties of tumor neoepitopes in humans. Cancer Immunol Res. 2:522–529
Bindea G, Mlecnik B, Tosolini M et al (2013) Spatiotemporal dynamics of intratumoral immune cells reveal the immune landscape in human cancer. Immunity 39:782–795
Galon J, Costes A, Sanchez-Cabo F et al (2006) Type, density, and location of immune cells within human colorectal tumors predict clinical outcome. Science 313:1960–1964
Charoentong P, Angelova M, Efremova M, Gallasch R, Hackl H, Galon J, Trajanoski Z (2012) Bioinformatics for cancer immunology and immunotherapy. Cancer Immunol Immunother 61:1885–1903
Volonte A, Di Tomaso T, Spinelli M et al (2014) Cancer-initiating cells from colorectal cancer patients escape from T cell-mediated immunosurveillance in vitro through membrane-bound IL-4. J Immunol. 192:523–532
Acknowledgments
Supported by Associazione Italiana per la Ricerca sul Cancro-AIRC to G. Casorati, G.Parmiani and P.Dellabona.
Conflict of interest
The authors declare no conflict of interest.
Author information
Authors and Affiliations
Corresponding authors
Additional information
This paper is a Focussed Research Review based on a presentation given at the Eleventh Meeting of the Network Italiano per la Bioterapia dei Tumori (NIBIT) on Cancer Bio-Immunotherapy, held in Siena, Italy, 17th–19th October 2013. It is part of a CII series of Focussed Research Reviews and meeting report.
Rights and permissions
About this article
Cite this article
Trajanoski, Z., Maccalli, C., Mennonna, D. et al. Somatically mutated tumor antigens in the quest for a more efficacious patient-oriented immunotherapy of cancer. Cancer Immunol Immunother 64, 99–104 (2015). https://doi.org/10.1007/s00262-014-1599-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00262-014-1599-7