Abstract
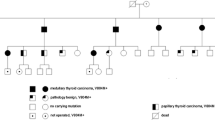
RET mutations play an important role in the development of human neuroendocrine tumors. The prevalence of the RET polymorphism G691S of exon 11 is higher in patients with medullary thyroid carcinoma (MTC) as compared to the general population. A weak association between RET polymorphisms and sporadic papillary thyroid carcinoma (PTC) has also been described. We hereby describe the association of MTC, bronchial carcinoid tumor, and PTC in a familial setting. A 75-yr-old woman developed MTC 7 yr after successful treatment of a bronchial carcinoid. Serum calcitonin was 12.9 pg/ml with a peak response to pentagastrin (151.0 pg/ml). The patient underwent total thyroidectomy and a genetic mutational analysis of the RET gene. Histological evaluation confirmed MTC with no evidence of lymph nodes involvement. After thyroidectomy serum calcitonin was <2.0 pg/ml. A germline missense mutation at codon 691 in exon 11 of the RET gene was found. The mutational analysis was extended to the patient’s offspring, and her daughter was found to bear the G691S polymorphism of RET. Wild type RET gene was found in the son. The daughter, who showed a nodular goiter, autoimmune thyroiditis and normal serum calcitonin, also underwent thyroidectomy. Histologic examination of the thyroid revealed an incidental PTC. This is the first description of a bronchial carcinoid tumor occurring in association with MTC. The occurrence of apparently unrelated NET in the same subject, or within a family, should be regarded as a challenge for deeper investigations into the possible oncogenic role of this genetic alteration.
Similar content being viewed by others
References
Oberg K. Carcinoid tumors, carcinoid syndrome and related disorders. In: Larsen PR, Kronenberg HM, Melmed S, Polonski KS, eds. Williams textbook of endocrinology. 10th ed. Philadelphia: WB Saunders. 2003, 1857–76.
Modlin IM, Lye KD, Kidd M. A 5-decade analysis of 13,715 carcinoid tumors. Cancer 2003, 97: 934–59.
Fialkowski EA, MoleyJF. Current approaches to medullary thyroid carcinoma, sporadic and familial. J Surg Oncol 2006, 94: 737–47.
Murakumo Y, Jijiwa M, Asai N, Ichihara M, Takahashi M. RET and neuroendocrine tumors. Pituitary 2006, 9: 179–92.
Plaza-Menacho I, Burzynski GM, de Groot JW, Eggen BJ, Hofstra RM. Current concepts in RET-related genetics, signaling and therapeutics. Trends Genet 2006, 22: 627–36.
De Groot JW, Links TP, Plukker JT, Lips CJ, Hofstra RM. RET as a diagnostic and therapeutic target in sporadic and hereditary endocrine tumors. Endocr Rev 2006, 27: 535–60.
Arighi E, Borrello MG, Sariola H. RET tyrosine kinase signaling in development and cancer. Cytokine Growth Factor Rev 2005, 16: 441–67.
Elisei R, Cosci B, Romei C, et al. Prognostic significance of somatic RET oncogene mutations in sporadic medullary thyroid cancer: a 10-year follow-up study. J Clin Endocrinol Metab 2008, 93: 682–7.
Robledo M, Gil L, Pollán M, et al. Polymorphisms G691S/S904S of RET as genetic modifiers of MEN 2A. Cancer Res 2003, 63: 1814–7.
Pacini F, Fontanelli M, Fugazzola L, et al. Routine measurement of serum calcitonin in nodular thyroid diseases allows the preoperative diagnosis of unsuspected sporadic medullary thyroid carcinoma. J Clin Endocrinol Metab 1994, 78: 826–9.
Boi F, Maurelli I, Pinna G, et al. Calcitonin measurement in washout fluid from fine needle aspiration of neck masses in patients with primary and metastatic medullary thyroid carcinoma. J Clin Endocrinol Metab 2007, 92: 2115–8.
Sachithanandan N, Harle RA, Burgess JR. Bronchopulmonary carcinoid in multiple endocrine neoplasia type 1. Cancer 2005, 103: 509–15.
Ichihara M, Murakumo Y, Takahashi M. RET and neuroendocrine tumors. Cancer Lett 2004, 204: 197–211.
Santoro M, Carlomagno F, Melillo RM, Fusco A. Dysfunction of the RET receptor in human cancer. Cell Mol Life Sci 2004, 61: 2954–64.
Weber F, Eng C. Editorial: germline variants within RET: clinical utility or scientific playtoy? J Clin Endocrinol Metab 2005, 90: 6334–6.
Elisei R, Cosci B, Romei C, et al. RET exon 11 (G691S) polymorphism is significantly more frequent in sporadic medullar thyroid carcinoma than in the general population. J Clin Endocrinol Metab 2004, 89: 3579–84.
Gursoy A, Erdogan MF, Erdogan G. Significance of the RET proto-oncogene polymorphisms in Turkish sporadic medullary thyroid carcinoma patients. J Endocrinol Invest 2006, 29: 858–62.
Lönn S, Bhatti P, Alexander BH, et al. Papillary thyroid cancer and polymorphic variants in TSHR- and RET-related genes: a nested case-control study within a cohort of U.S. radiologic technologists. Cancer Epidemiol Biomarkers Prev 2007, 16: 174–7.
Lesueur F, Cebrian A, Robledo M, et al. Polymorphisms in RET and its coreceptors and ligands as genetic modifiers of multiple endocrine neoplasia type 2A. Cancer Res 2006, 66: 1177–80.
Sawai H, Okada Y, Kazanjian K, et al. The G691S RET polymorphism increases glial cell line-derived neurotrophic factor-induced pancreatic cancer cell invasion by amplifying mitogen-activated protein kinase signaling. Cancer Res 2005, 65: 11536–44.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Rotondi, M., Ercolino, T., Fonte, R. et al. Occurrence of medullary thyroid carcinoma, bronchial carcinoid tumor, and papillary thyroid carcinoma in a family bearing the RET G691S polymorphism. J Endocrinol Invest 32, 115–118 (2009). https://doi.org/10.1007/BF03345698
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/BF03345698